Abstract
Introduction: Metastasis-directed therapy (MDT) is a promising approach for recurrent or de novo oligo-metastatic castration-sensitive prostate cancer (omCSPC). The aim of this study was to evaluate the treatment approaches in omCSPC among German physicians. Methods: An anonymous online questionnaire was sent via survio.com to the members of the German Societies of Urology and Radiooncology. Results: Participants (n = 166; 33% urologists, 66% radiooncologists) define omCSPC as ≤3 (31%) or ≤4 (58%) metastases. Multimodal therapy consisting of local therapy of the primary tumor, MDT, and androgen deprivation therapy (ADT) was favored. For local therapy, radiotherapy was the preferred approach (radiotherapy: 84%, prostatectomy: 16%). Overall, 77% and 76% considered MDT as (very) highly important in synchronous and metachronous omCSPC, respectively. In total, 80% would complement MDT with time-limited ADT. Compared to urologists, radiooncologists more often include cases with ≥3 metastases (p = 0.006) and see a higher importance of radiotherapy (p = 0.023), a lower importance of prostatectomy (p < 0.001) as well as a higher importance of MDT (in de novo p = 0.038, in metachronous p = 0.010). Conclusion: MDT with time-limited ADT is a common treatment strategy in omCSPC. Especially in synchronous omCSPC, radiotherapy as local treatment for the primary is the preferred option rather than radical prostatectomy.
Introduction
Approximately 20% of prostate cancers (PCas) are present with locally advanced disease or evidence of metastases at initial diagnosis [1]. By using molecular imaging such as prostate-specific membrane antigen (PSMA)-positron emission tomography (PET)/computed tomography (CT), metastases can be detected earlier [2]. As a result, approximately 20 to 30% of patients with clinically localized high-risk PCa on conventional imaging actually have metastatic disease on PSMA imaging [2]. PSMA-PET imaging is also of great importance in the diagnosis of recurrent PCa after local treatment of the primary [3].
Metastatic PCa is currently no longer considered amenable to curative therapy; the median survival time is approximately 5 years [4‒8]. Treatment is based on androgen deprivation therapy (ADT) with intensification by an androgen receptor signaling inhibitor (ARSI) and/or taxane-based chemotherapy [9, 10]. Combination therapies with ADT and ARSI such as enzalutamide or apalutamide appear to have an advantage for overall survival, particularly in low-volume metastatic PCa [6, 8].
In 1995, Hellman and Weichselbaum [11] postulated the concept of oligo-metastatic cancers that could potentially be treated curatively by metastasis-directed therapy (MDT). In particular, patients with a limited number of metastases have a better prognosis and may benefit from local treatment of the primary [12] and/or its metastases [13‒21]. Nevertheless, patients with oligo-metastatic castration-sensitive PCa (omCSPC) are currently still treated with palliative intent. With the improvement of local treatment strategies for these patients, a cure might even be possible. So, radiotherapy (RTx) of the primary in combination with ADT showed an overall survival benefit compared to ADT monotherapy in the STAMPEDE trial in patients with a maximum of 3 metastases [12]. Currently, MDT is a promising experimental therapeutic approach for recurrent or de novo omCSPC. MDT is performed as stereotactic ablative body radiotherapy, as surgical metastasectomy or a combination of both. It can thus play a role in tumor control and disease progression. The aims of MDT or local therapy of the primary tumor in oligo-metastatic disease are to prolong overall survival, but also progression-free survival (PFS), and to delay the timing of ADT or therapy intensification or to delay castration resistance. Prospective studies in metachronous and synchronous omCSPC with small case numbers indicate effective long-term local control of the metastases. Thus, a delay in tumor progression and a postponement of ADT will occur. Data for a combined therapy of the primary by means of radical prostatectomy (RPx) or RTx with MDT and intensified ADT as well as their temporal limitation are pending. Moreover, there is no uniform definition for the term oligo-metastases with regard to the number of metastases, metastasis volume, or metastasis distribution [22]. The aim of this study was to evaluate the treatment approach for omCSPC among German physicians involved in the treatment of omCSPC.
Methods
Survey
A 35-item questionnaire was designed to collect demographic data and information on German physicians’ opinions regarding the treatment aspects in patients with metachronous or synchronous omCSPC. Mainly, physicians involved in the treatment of metastatic PCa were addressed. The questionnaire included questions about the diagnosis, definition, and treatment options in these patients. Moreover, the majority of questions aimed to explore the use of local treatment of the primary, MDT, and the use of systemic treatment. The questionnaire contained open questions, multiple choice questions, and certain questions that allowed respondents to “select all that apply.” Information was obtained on the respondents’ age, gender, practice region, urban area, practice type, level of training, years in practice, number of treated patients with (metastatic) PCa and use of local treatment of the primary, MDT, and time-limited systemic treatment in daily practice.
Study Design
A link to the survey together with a personal invitation from the German Prostate Cancer Consortium (DPKK) was sent three times with a time interval of 6 weeks through email to all members of the German Society of Urology (DGU) and German Society of Radiooncology (DGRO). With over 6,500 (DGU) and 2,389 (DGRO) members, these societies represent two large medical societies in Germany, with a heterogeneous member population of clinicians and urologic and radiooncologic practitioners in communal and academic hospitals or in outpatient offices. Approximately 9,000 members received the email invitation. The responses were collected in an SPSS spreadsheet in an anonymous fashion. Statistical analyses were performed using SPSS version 26.0 (IBM, Mount Kisco, NY, USA). Respondents’ age, gender, practice region, urban area, practice type, level of training, experience in urology practice, and experience in treating PCa patients were used in regression analyses. For significant associations identified in the univariate regression analyses, all factors were identified that changed the calculated odds ratio >10%, and these were included in the respective multivariate model. Statistical significance was defined as p < 0.05.
Results
A total of 166 responses were received. The characteristics of the participants are shown in detail in Table 1. Briefly, the median age of participants was 51 years (interquartile range 40–59 years) and 62% were male. The majority of participants were board-certified physicians from different disciplines (33% urologists, 66% radiooncologists) with a working experience of ≥10 years. Two-thirds of participants live and work in metropolitan areas. Here, 25% work at a university hospital, 47% in their own outpatient medical practice, and the remainder in academic (12%) or communal hospitals (16%). On average, the participants see 50–100 patients with any stage PCa, 11–50 patients with metastatic PCa, and 1–10 patients with omCSPC per 3 months.
Characteristics of survey participants
| Parameter . | n = 166 . |
|---|---|
| Age (median, IQR), years | 51 (40–59) |
| Gender, n (%) | |
| Male | 102 (62) |
| Female | 62 (37) |
| Divers | 1 (1) |
| Certified physicians, n (%) | 155 (93) |
| Specialization, n (%) | |
| Urologist | 53 (33) |
| Radiooncologist | 109 (66) |
| Oncologist | 1 (1) |
| Regions, n (%) | |
| Western Germany | 133 (83) |
| Eastern Germany | 27 (17) |
| Area, n (%) | |
| Rural-suburban (<20,000 citizens) | 10 (6) |
| Urban (20,000–100,000 citizens) | 45 (27) |
| Metropolitan (>100,000 citizens) | 110 (66) |
| Practice type, n (%) | |
| University hospital | 41 (25) |
| Academic hospital | 20 (12) |
| Communal hospital | 26 (16) |
| Medical practice | 78 (47) |
| Work experience, n (%) | |
| 1–5 years | 7 (4) |
| 6–10 years | 20 (12) |
| 11–20 years | 48 (29) |
| >20 years | 90 (65) |
| Patients with PCa/quarter, n (%) | |
| <50 | 60 (36) |
| 50–100 | 71 (43) |
| 101–200 | 20 (12) |
| >200 | 12 (7) |
| Patients with metastasized PCa/quarter, n (%) | |
| <10 | 31 (19) |
| 11–20 | 65 (39) |
| 21–50 | 51 (31) |
| 51–100 | 12 (7) |
| >100 | 4 (2.4) |
| Patients with oligo-mCSPC/quarter, n (%) | |
| 1–5 | 88 (53) |
| 6–10 | 61 (37) |
| 11–20 | 11 (6.6) |
| >20 | 5 (3) |
| Parameter . | n = 166 . |
|---|---|
| Age (median, IQR), years | 51 (40–59) |
| Gender, n (%) | |
| Male | 102 (62) |
| Female | 62 (37) |
| Divers | 1 (1) |
| Certified physicians, n (%) | 155 (93) |
| Specialization, n (%) | |
| Urologist | 53 (33) |
| Radiooncologist | 109 (66) |
| Oncologist | 1 (1) |
| Regions, n (%) | |
| Western Germany | 133 (83) |
| Eastern Germany | 27 (17) |
| Area, n (%) | |
| Rural-suburban (<20,000 citizens) | 10 (6) |
| Urban (20,000–100,000 citizens) | 45 (27) |
| Metropolitan (>100,000 citizens) | 110 (66) |
| Practice type, n (%) | |
| University hospital | 41 (25) |
| Academic hospital | 20 (12) |
| Communal hospital | 26 (16) |
| Medical practice | 78 (47) |
| Work experience, n (%) | |
| 1–5 years | 7 (4) |
| 6–10 years | 20 (12) |
| 11–20 years | 48 (29) |
| >20 years | 90 (65) |
| Patients with PCa/quarter, n (%) | |
| <50 | 60 (36) |
| 50–100 | 71 (43) |
| 101–200 | 20 (12) |
| >200 | 12 (7) |
| Patients with metastasized PCa/quarter, n (%) | |
| <10 | 31 (19) |
| 11–20 | 65 (39) |
| 21–50 | 51 (31) |
| 51–100 | 12 (7) |
| >100 | 4 (2.4) |
| Patients with oligo-mCSPC/quarter, n (%) | |
| 1–5 | 88 (53) |
| 6–10 | 61 (37) |
| 11–20 | 11 (6.6) |
| >20 | 5 (3) |
IQR, interquartile range; mCSPC, metastatic castration-sensitive prostate cancer; PCa, prostate cancer.
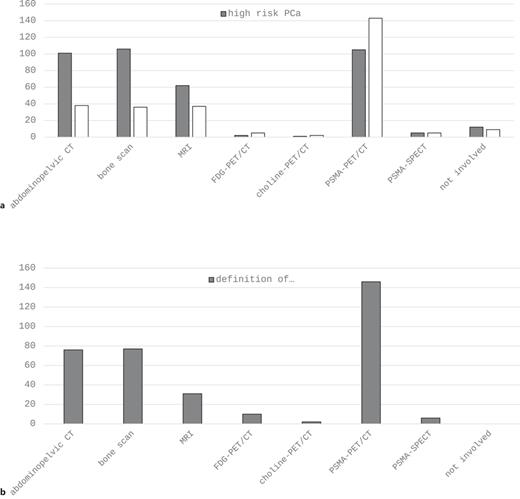
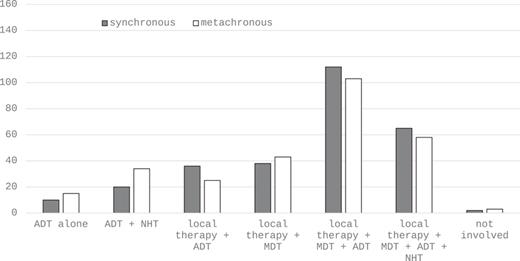
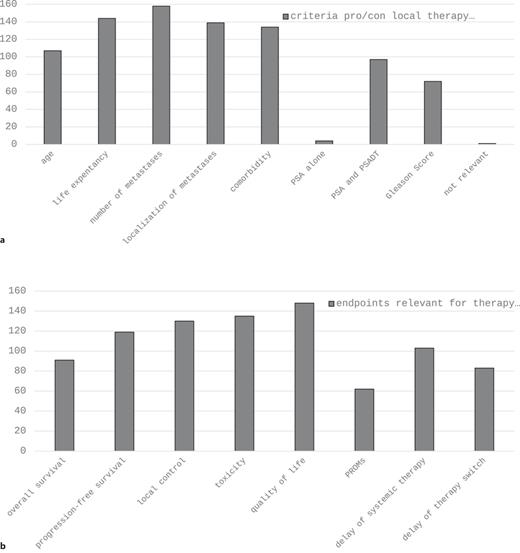
Concerning imaging in the primary staging of high-risk PCa, abdominopelvic CT, bone scan, and PSMA-PET/CT demonstrate the three most widely used modalities with comparable mentions (Fig. 1a). In the setting of biochemical recurrence, PSMA-PET/CT is by far the most frequently applied method (Fig. 1b). About 90% of participants define omCSPC as the presence of ≤3 (31%) or ≤4 (58%) metastases in imaging studies (Table 2). As far as treatment of omCSPC is concerned, most participants favored an aggressive multimodal combination therapy consisting of local therapy of the primary tumor, MDT, and ADT (Fig. 2). Looking more detailed on local therapy in de novo omCSPC, the majority considered RTx of the prostate to be highly important (58%) or very highly important (17%). This was not the case for RPx (highly important: 12.7%, very highly important: 4.2%). Accordingly, RTx was clearly the preferred approach for local therapy (RTx: 84%, prostatectomy: 13%). Aside from treatment of the primary tumor, 77% and 76% considered MDT as highly or very highly important in synchronous and metachronous omCSPC, respectively (Table 2). About half of the participants would even consider MDT for visceral metastases. Looking at prerequisites to evaluate patients with omCSPC as eligible for local therapy +/− MDT, 65% of participants would like their patients to have a life expectancy of at least 5 years. Besides life expectancy, the most important factors influencing the choice of treatment in those patients also include age, number, and localization of metastases as well as comorbidities (Fig. 3a). Asking for the most relevant clinical endpoints in patients with omCSPC, quality of life, toxicity, local tumor control, and PFS were most frequently mentioned followed by delay of systemic therapy and overall survival (Fig. 3b). When applying local therapy and MDT in omCSPC, most participants would complement these with the use of ADT (80%). However, in synchronous as well as metachronous omCSPC, the majority would limit the use of ADT for a duration of maximum 2 years (Table 2).
a Imaging performed in newly diagnosed high-risk PCa and biochemical recurrence. b Definition of oligo-metastatic PCa by imaging. CT, computed tomography; FDG, fluorodeoxyglucose; MRI, magnetic resonance imaging; PCa, prostate cancer; PET, positron emission tomography; PSMA, prostate-specific membrane antigen; SPECT, single photon emission computed tomography.
a Imaging performed in newly diagnosed high-risk PCa and biochemical recurrence. b Definition of oligo-metastatic PCa by imaging. CT, computed tomography; FDG, fluorodeoxyglucose; MRI, magnetic resonance imaging; PCa, prostate cancer; PET, positron emission tomography; PSMA, prostate-specific membrane antigen; SPECT, single photon emission computed tomography.
Questions on the management of oligo-metastatic PCa
| Definition of oligo-mCSPC | |
| 1 metastasis | 1 (1%) |
| ≤3 metastases | 52 (31%) |
| ≤4 metastases | 97 (58%) |
| ≤8 metastases | 9 (5.4%) |
| ≤10 metastases | 2 (1.2%) |
| Not important for therapy decision | 2 (1.2%) |
| Importance of RTx of the primary in de novo oligo-mHSPC | |
| Low | 3 (1.8%) |
| Moderate | 26 (15.7%) |
| Equivocal | 10 (6%) |
| High | 96 (58%) |
| Very high | 27 (17%) |
| Preferred local therapy of the primary | |
| RTx | 139 (84%) |
| Prostatectomy | 21 (13%) |
| Others | 1 (1%) |
| I do not know | 2 (1.2%) |
| Importance of prostatectomy of the primary in de novo oligo-mHSPC | |
| Low | 63 (38%) |
| Moderate | 52 (31%) |
| Equivocal | 20 (12%) |
| High | 21 (12.7%) |
| Very high | 7 (4.2%) |
| Importance of MDT in de novo oligo-mHSPC | |
| Low | 3 (2%) |
| Moderate | 29 (12%) |
| Equivocal | 12 (7%) |
| High | 96 (58%) |
| Very high | 31 (19%) |
| Importance of MDT in metachronous oligo-mHSPC | |
| Low | 1 (1%) |
| Moderate | 24 (15%) |
| Equivocal | 11 (7%) |
| High | 100 (60%) |
| Very high | 27 (16%) |
| Importance of RTx of the primary and MDT in de novo oligo-mHSPC | |
| Low | 2 (1%) |
| Moderate | 11 (7%) |
| Equivocal | 13 (8%) |
| High | 102 (61%) |
| Very high | 34 (21%) |
| Importance of prostatectomy of the primary and MDT in de novo oligo-mHSPC | |
| Low | 62 (37%) |
| Moderate | 49 (30%) |
| Equivocal | 25 (15%) |
| High | 20 (12%) |
| Very high | 6 (4%) |
| Would you consider local treatment of visceral metastases in oligo-mCSPC? | |
| No | 75 (45%) |
| Yes | 86 (52%) |
| Minimum life expectancy to consider local therapy +/− MDT in oligo-mHSPC | |
| >5 years | 108 (65%) |
| >10 years | 28 (17%) |
| Not relevant | 21 (13%) |
| I do not know | 5 (3%) |
| Would you use phoenix criteria for definition of recurrence after MDT + RTx of the primary? | |
| No | 66 (40%) |
| Yes | 64 (39%) |
| I do not know | 32 (19%) |
| Would you use a PSA >0.2 ng/mL cut-off for definition of recurrence after MDT + prostatectomy? | |
| No | 46 (28%) |
| Yes | 87 (52%) |
| I do not know | 0 |
| Additional treatment with ADT parallel to MDT and local therapy in oligo-mHSPC? | |
| Yes | 127 (80%) |
| No | 36 (22%) |
| Time limitation of ADT parallel to MDT and local therapy in de novo oligo-mHSPC? | |
| No | 36 (22%) |
| Yes | 118 (71%) |
| Not carried out | 8 (5%) |
| Duration of ADT parallel to MDT and local therapy in de novo oligo-mHSPC | |
| 6 months | 17 (10%) |
| 1 year | 15 (9%) |
| 2 years | 91 (55%) |
| 5 years | 5 (3%) |
| Not limited | 23 (14%) |
| No ADT is carried out | 9 (5%) |
| Time limitation of ADT parallel to MDT in metachronous oligo-mHSPC? | |
| No | 49 (30%) |
| Yes | 93 (56%) |
| Not carried out | 17 (10%) |
| Duration of ADT parallel to MDT in metachronous oligo-mHSPC | |
| 6 months | 19 (11%) |
| 1 year | 11 (7%) |
| 2 years | 70 (42%) |
| 5 years | 2 (1%) |
| Not limited | 38 (23%) |
| No ADT is carried out | 17 (10%) |
| Definition of oligo-mCSPC | |
| 1 metastasis | 1 (1%) |
| ≤3 metastases | 52 (31%) |
| ≤4 metastases | 97 (58%) |
| ≤8 metastases | 9 (5.4%) |
| ≤10 metastases | 2 (1.2%) |
| Not important for therapy decision | 2 (1.2%) |
| Importance of RTx of the primary in de novo oligo-mHSPC | |
| Low | 3 (1.8%) |
| Moderate | 26 (15.7%) |
| Equivocal | 10 (6%) |
| High | 96 (58%) |
| Very high | 27 (17%) |
| Preferred local therapy of the primary | |
| RTx | 139 (84%) |
| Prostatectomy | 21 (13%) |
| Others | 1 (1%) |
| I do not know | 2 (1.2%) |
| Importance of prostatectomy of the primary in de novo oligo-mHSPC | |
| Low | 63 (38%) |
| Moderate | 52 (31%) |
| Equivocal | 20 (12%) |
| High | 21 (12.7%) |
| Very high | 7 (4.2%) |
| Importance of MDT in de novo oligo-mHSPC | |
| Low | 3 (2%) |
| Moderate | 29 (12%) |
| Equivocal | 12 (7%) |
| High | 96 (58%) |
| Very high | 31 (19%) |
| Importance of MDT in metachronous oligo-mHSPC | |
| Low | 1 (1%) |
| Moderate | 24 (15%) |
| Equivocal | 11 (7%) |
| High | 100 (60%) |
| Very high | 27 (16%) |
| Importance of RTx of the primary and MDT in de novo oligo-mHSPC | |
| Low | 2 (1%) |
| Moderate | 11 (7%) |
| Equivocal | 13 (8%) |
| High | 102 (61%) |
| Very high | 34 (21%) |
| Importance of prostatectomy of the primary and MDT in de novo oligo-mHSPC | |
| Low | 62 (37%) |
| Moderate | 49 (30%) |
| Equivocal | 25 (15%) |
| High | 20 (12%) |
| Very high | 6 (4%) |
| Would you consider local treatment of visceral metastases in oligo-mCSPC? | |
| No | 75 (45%) |
| Yes | 86 (52%) |
| Minimum life expectancy to consider local therapy +/− MDT in oligo-mHSPC | |
| >5 years | 108 (65%) |
| >10 years | 28 (17%) |
| Not relevant | 21 (13%) |
| I do not know | 5 (3%) |
| Would you use phoenix criteria for definition of recurrence after MDT + RTx of the primary? | |
| No | 66 (40%) |
| Yes | 64 (39%) |
| I do not know | 32 (19%) |
| Would you use a PSA >0.2 ng/mL cut-off for definition of recurrence after MDT + prostatectomy? | |
| No | 46 (28%) |
| Yes | 87 (52%) |
| I do not know | 0 |
| Additional treatment with ADT parallel to MDT and local therapy in oligo-mHSPC? | |
| Yes | 127 (80%) |
| No | 36 (22%) |
| Time limitation of ADT parallel to MDT and local therapy in de novo oligo-mHSPC? | |
| No | 36 (22%) |
| Yes | 118 (71%) |
| Not carried out | 8 (5%) |
| Duration of ADT parallel to MDT and local therapy in de novo oligo-mHSPC | |
| 6 months | 17 (10%) |
| 1 year | 15 (9%) |
| 2 years | 91 (55%) |
| 5 years | 5 (3%) |
| Not limited | 23 (14%) |
| No ADT is carried out | 9 (5%) |
| Time limitation of ADT parallel to MDT in metachronous oligo-mHSPC? | |
| No | 49 (30%) |
| Yes | 93 (56%) |
| Not carried out | 17 (10%) |
| Duration of ADT parallel to MDT in metachronous oligo-mHSPC | |
| 6 months | 19 (11%) |
| 1 year | 11 (7%) |
| 2 years | 70 (42%) |
| 5 years | 2 (1%) |
| Not limited | 38 (23%) |
| No ADT is carried out | 17 (10%) |
ADT, androgen deprivation therapy; MDT, metastasis-directed therapy; mCSPC, metastatic castration-sensitive prostate cancer; PCa, prostate cancer; PSA, prostate-specific antigen.
Treatment approaches in synchronous (dark bars) and metachronous (light bars) oligo-metastatic PCa. ADT, androgen deprivation therapy; MDT, metastasis-directed therapy; NHT, new hormonal therapy.
Treatment approaches in synchronous (dark bars) and metachronous (light bars) oligo-metastatic PCa. ADT, androgen deprivation therapy; MDT, metastasis-directed therapy; NHT, new hormonal therapy.
a Criteria considered for the choice pro/con local therapy +/− MDT. b Endpoints relevant for therapy decision in patients with oligo-metastatic PCa. MDT, metastasis-directed therapy; PROMs, patient-reported outcome measurements; PSA, prostate-specific antigen; PSADT, PSA doubling time.
a Criteria considered for the choice pro/con local therapy +/− MDT. b Endpoints relevant for therapy decision in patients with oligo-metastatic PCa. MDT, metastasis-directed therapy; PROMs, patient-reported outcome measurements; PSA, prostate-specific antigen; PSADT, PSA doubling time.
While the place of work (university hospital vs. nonuniversity hospital) seemed to be not of utmost importance for the management of patients with omCSPC, there were significant differences in many points depending on the professions (urologists vs. radiooncologists; Table 3). Specifically, compared to urologists, radiooncologists more often include cases with more than 3 metastases in the group of omCSPC (p = 0.006), see a higher importance of RTx (p = 0.023), a lower importance of RPx (p < 0.001) in de novo omCSPC, a higher importance of MDT in de novo (p = 0.038), and metachronous (p = 0.010) omCSPC, and more often consider MDT also for visceral metastases (p < 0.001).
Difference in the management of oligo-metastatic PCa according to profession (urologists/urooncologists vs. radiooncologists) and practice type (university hospital vs. other practice types)
| . | Urologist . | Radiooncologist . | p value . | University hospital . | Nonuniversity hospital . | p value . |
|---|---|---|---|---|---|---|
| Definition of oligo-mCSPC (>3 vs. ≤3 metastases) | 28/53 (53%) | 79/106 (75%) | p = 0.006 | 31/40 (78%) | 77/121 (64%) | p = 0.106 |
| Importance of RTx of the primary in de novo oligo-mCSPC (high or very high) | 34/52 (65%) | 89/109 (82%) | p = 0.023 | 29/40 (73%) | 95/123 (77%) | p = 0.542 |
| Importance of prostatectomy in de novo oligo-mCSPC (high or very high) | 21/52 (40%) | 7/109 (6%) | p < 0.001 | 11/40 (28%) | 17/123 (14%) | p = 0.046 |
| Importance of MDT in de novo oligo-mCSPC (high or very high) | 36/52 (69%) | 91/109 (83%) | p = 0.038 | 31/40 (78%) | 97/123 (79%) | p = 0.855 |
| Importance of MDT in metachronous oligo-mCSPC (high or very high) | 34/52 (65%) | 91/109 (83%) | p = 0.010 | 35/40 (88%) | 92/123 (75%) | p = 0.092 |
| Importance of RTx of the primary and MDT in de novo oligo-mCSPC (high or very high) | 40/52 (77%) | 95/108 (88%) | p = 0.072 | 35/40 (88%) | 91/122 (75%) | p = 0.481 |
| Importance of prostatectomy and MDT in de novo oligo-mCSPC (high or very high) | 9/52 (17%) | 17/108 (16%) | p = 0.801 | 4/40 (10%) | 22/122 (18%) | p = 0.230 |
| Importance of local treatment of visceral metastases | 14/52 (27%) | 72/107 (67%) | p < 0.001 | 18/40 (45%) | 68/121 (56%) | p = 0.218 |
| Importance of time-limited ADT in de novo oligo-mCSPC (high or very high) | 33/48 (69%) | 84/104 (81%) | p = 0.102 | 33/40 (83%) | 85/114 (75%) | p = 0.307 |
| Importance of time-limited ADT in metachronous oligo-mCSPC (high or very high) | 24/44 (55%) | 68/97 (70%) | p = 0.072 | 28/37 (76%) | 65/105 (62%) | p = 0.130 |
| . | Urologist . | Radiooncologist . | p value . | University hospital . | Nonuniversity hospital . | p value . |
|---|---|---|---|---|---|---|
| Definition of oligo-mCSPC (>3 vs. ≤3 metastases) | 28/53 (53%) | 79/106 (75%) | p = 0.006 | 31/40 (78%) | 77/121 (64%) | p = 0.106 |
| Importance of RTx of the primary in de novo oligo-mCSPC (high or very high) | 34/52 (65%) | 89/109 (82%) | p = 0.023 | 29/40 (73%) | 95/123 (77%) | p = 0.542 |
| Importance of prostatectomy in de novo oligo-mCSPC (high or very high) | 21/52 (40%) | 7/109 (6%) | p < 0.001 | 11/40 (28%) | 17/123 (14%) | p = 0.046 |
| Importance of MDT in de novo oligo-mCSPC (high or very high) | 36/52 (69%) | 91/109 (83%) | p = 0.038 | 31/40 (78%) | 97/123 (79%) | p = 0.855 |
| Importance of MDT in metachronous oligo-mCSPC (high or very high) | 34/52 (65%) | 91/109 (83%) | p = 0.010 | 35/40 (88%) | 92/123 (75%) | p = 0.092 |
| Importance of RTx of the primary and MDT in de novo oligo-mCSPC (high or very high) | 40/52 (77%) | 95/108 (88%) | p = 0.072 | 35/40 (88%) | 91/122 (75%) | p = 0.481 |
| Importance of prostatectomy and MDT in de novo oligo-mCSPC (high or very high) | 9/52 (17%) | 17/108 (16%) | p = 0.801 | 4/40 (10%) | 22/122 (18%) | p = 0.230 |
| Importance of local treatment of visceral metastases | 14/52 (27%) | 72/107 (67%) | p < 0.001 | 18/40 (45%) | 68/121 (56%) | p = 0.218 |
| Importance of time-limited ADT in de novo oligo-mCSPC (high or very high) | 33/48 (69%) | 84/104 (81%) | p = 0.102 | 33/40 (83%) | 85/114 (75%) | p = 0.307 |
| Importance of time-limited ADT in metachronous oligo-mCSPC (high or very high) | 24/44 (55%) | 68/97 (70%) | p = 0.072 | 28/37 (76%) | 65/105 (62%) | p = 0.130 |
Two-sided chi-square tests were used to calculate statistical significance.
ADT, androgen deprivation therapy; mCSPC, metastatic castration-sensitive prostate cancer.
Discussion
MDT for synchronous and oligo-metastatic omCSPC is a modern treatment concept. It is offered to patients for a good local control rate and to delay further treatment intensification or even in a potentially curative intent. Hence, the aim of local therapy is to delay tumor progression and reduce or delay the need for ADT. However, prospective, randomized studies for this therapy concept are still pending, so it has not yet been strongly recommended in national and international guidelines. Only for local RTx of the primary combined with ADT, evidence is based on data from the STAMPEDE study [12]. Nevertheless, MDT is widely offered especially in high-volume centers. With this survey, we were able to show that local treatment of metastases is a widely used treatment concept for both synchronous and metachronous omCSPC. Only a small proportion of urologists and radiotherapists, who responded in this survey, do not offer this to their patients.
The majority of responders are working in outpatient offices, only one third worked in a university hospital or in an academic setting. However, the working environment did not appear to have any influence on the use of MDT. The majority of responders considered MDT as highly or very highly important in synchronous and metachronous omCSPC.
In synchronous metastatic omCSPC, both urologists and radiotherapists attach greater importance to local therapy of the primary using RTx. Only a small proportion of those surveyed attached great importance to RPx for the treatment of the primary in the omCSPC setting.
The most relevant clinical endpoints in patients with omCSPC, quality of life, toxicity, local tumor control, and PFS were most frequently mentioned followed by delay of systemic therapy and overall survival. The arm H of the STAMPEDE study showed that treatment of the primary with RTx and ADT is superior to ADT monotherapy in omCSPC with up to 3 metastases for the endpoint overall survival. These data evoke that local control of the primary showed treatment effects. However, an additional MDT to the metastases was not performed in that trial [12]. Data for the benefit of local tumor control by RPx in the metastatic setting are limited. A retrospective propensity matched cohort analysis of 4,000 patients with omCSPC from the SEER database showed a slightly tumor-specific survival benefit for patients undergoing RPx compared to RTx of the primary [23].
When applying local therapy and MDT in omCSPC, most responders would complement these with the use of ADT (80%). This is concurrent with the ESTRO-ACROP Delphi consensus conference where 64% and 76% of participants regarded systemic treatment as baseline treatment additional to MDT in the metachronous and synchronous omCSPC, respectively [24]. Again, clinical data to omit ADT in this setting are sparse. Phase III studies investigating ADT combined with ARSI like apalutamide or enzalutamide in synchronous and metachronous omCSPC showed a significant survival benefit compared to ADT monotherapy also in patients with low metastatic burden [6, 8]. Especially in the metachronous setting, clinical phase II trials with only small numbers of included patients showed a delay of prostate-specific antigen-PFS, ADT initiation with MDT, and a good local control versus observation [14, 18, 21]. However, data for overall survival are still pending. Kroeze et al. [25] demonstrated that additional long-term (≥6 months) ADT showed a benefit for PFS in a retrospective study including 305 patients. However, prospective data investigating MDT and additional (time-limited) ADT or ADT in combination with ARSI are sparse. The phase II EXTEND-2 trial investigating 87 patients with ADT monotherapy and MDT plus ADT demonstrated a longer PFS with a hazard ratio of 0.25 (0.12–0.55) for MDT plus ADT versus ADT monotherapy [26]. The one-arm SATURN trial presented a 12-month PFS of 69% in 28 patients treated with MDT in combination with ADT plus abiraterone and apalutamide [27]. The SPARKLE trial investigating MDT alone, MDT with 1 month of ADT, and MDT with 6 months of ADT plus enzalutamide including >800 patients will give further information about the need of ADT in this setting [28].
Radiooncologists more often include cases with ≥3 metastases in the group of omCSPC, see a higher importance of RTx and a lower importance of RPx in de novo omCSPC as well as a higher importance of MDT in de novo and metachronous omCSPC, and more often consider MDT even for visceral metastases. In the Delphi consensus including only radiooncologist, the MDT approach was considered until 5 metastases. However visceral metastases were only considered in selected cases [24]. Rogowski et al. [29] described in a survey among German radiooncologists in 2022 that the majority define oligo-metastatic disease by PSMA-PET imaging. In the metachronous omCSPC, 63% of radiooncologists aim to defer systemic treatment. In the omCSPC, MDT is performed mainly in bone or lymph node metastasis [29]. Moreover, the case-based survey by Yoo et al. [30] demonstrated a high consensus among radiooncologists in the indication and treatment in patients with omCSPC.
Our study has its limitations. First, the low proportion of responses to the study may mean that the answers are not entirely representative. However, we surveyed both radiooncologists and urologists from the specialist societies. We are aware that more radiooncologists were involved in this survey than urologists. Therefore, the use of MDT also in a higher number of metastasis might be favored and represent a bias in our survey. However, the proportion of the working environment also appears representative compared to the distribution in patient care since especially in MDT by stereotactic RTx radiooncologists are more frequently involved in the treatment. Furthermore, there is a possible risk of bias due to the slightly higher proportion of radiooncologists and the relatively high proportion of respondents from the university environment. Nevertheless, we believe that our study is important to understand the current treatment strategies aside from the lack of strong recommendations for MDT and strategies for the implementation of systemic therapy in the guidelines for this patient group.
In conclusion, MDT with a time-limited ADT for 2 years is a common therapy strategy for patients with omHSPC for urologists as well as for radiooncologists. Especially in synchronous omHSPC, RTx as local treatment for the primary is the preferred option rather than RPx.
Acknowledgments
We thank Olaf Kurpick for preparation of the questionnaire and for support with the technical details.
Statement of Ethics
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional research committee and with the 1964 Helsinki Declaration and its later amendments or comparable standards. Ethical approval was not required for this study in accordance with local/national guidelines. Written informed consent to participate was not directly obtained but inferred by completion of the questionnaire/participation in the interview. This study does not contain any studies with animals.
Conflict of Interest Statement
Angelika Borkowetz and Bernd Wullich were members of the journal’s Editorial Board at the time of submission. The remaining authors have no conflicts of interest to declare.
Funding Sources
This study was not supported by any sponsor or funder.
Author Contributions
A.B.: study concept, data collection, data analysis, data interpretation, drafting of the manuscript, and revision of the manuscript. B.W. and M.S.: conception and design, supervision, and revision of the manuscript. N.S.-H.: conception and design, and revision of the manuscript. J.L.: study concept, data interpretation, drafting of the manuscript, and revision of the manuscript.
Data Availability Statement
The data that support the findings of this study are not publicly available due to containing information that could compromise the privacy of research participants but are available from the corresponding author.