Abstract
Introduction: Ablation therapy (AT) and partial nephrectomy (PN) are primary treatment options for renal cell carcinoma (RCC). This study aimed to perform a systematic review and meta-analysis to compare the efficacy and safety of AT and PN in treating RCC. Methods: This study was performed in accordance with the PRISMA guidelines. A comprehensive literature search of PubMed, Embase, Cochrane Library, and Web of Science was conducted for studies published up to February 1, 2025. Statistical analyses were performed using Stata16 software. Results: A total of 32 studies involving 6,030 patients were included. The analysis demonstrated that AT was associated with significantly shorter operative time (OT), less estimated blood loss (EBL), a shorter length of hospital stay (LOS), and a lower overall complication rate (CR) compared to PN. Compared with AT, PN has more advantages in overall survival and recurrence-free survival. There were no statistically significant differences between the two interventions in cancer-specific survival. Conclusion: For selected patients with RCC, AT represents a minimally invasive alternative that offers advantages over PN in perioperative outcomes, including reduced OT, EBL, LOS, and CR, while preserving renal function. However, no significant differences were found in long-term oncological survival outcomes.
Introduction
The prevalence of renal cell carcinoma (RCC) is rising at an annual rate of 3–5%, and it is estimated that approximately 140,000 fatalities per year are attributed to renal cancer [1, 2]. The therapeutic strategies for RCC encompass radical nephrectomy, partial nephrectomy (PN), and active surveillance [3]. PN serves as the primary treatment for localized RCC due to its ability to effectively preserve renal function [4]. However, for patients who are unsuitable for surgical intervention, ablation therapy (AT) has emerged as a valuable alternative. These techniques can precisely destroy tumor tissues without open surgery, offering a less invasive approach with improved patient tolerance [2].
In recent years, significant advancements have been made in AT for RCC. Technologically, AT now encompasses multiple modalities including cryoablation (CA), radiofrequency ablation (RFA), and microwave ablation (MWA) [5]. In terms of surgical access, there has been a shift from traditional laparoscopic ablation therapy (LAT) toward the less invasive percutaneous ablation therapy (PAT) [6, 7]. A recent meta-analysis involving 2,011 patients compared CA and PN for RCC and found comparable perioperative outcomes and renal function preservation between the two techniques [8]. However, another study indicated that patients selected for RFA, CA, or MWA tended to be older and have more comorbidities than those undergoing PN. Although cancer-specific survival (CSS) was similar across groups, the rate of local recurrence was consistently higher after any ablative treatment compared to PN [9].
There remains considerable controversy in the current literature. Some meta-analyses are limited by an insufficient number of included studies [10], while others compare ablation technologies (such as RFA, CA, and MWA) with PN without adequately accounting for differences between ablation modalities or distinguishing between laparoscopic and percutaneous approaches [11]. Therefore, this study aimed to systematically review a broad range of clinical evidence to clarify the safety and efficacy profiles of various ablation techniques, different surgical access routes, and AT in the management of RCC.
Methods
In February 2025, we conducted a systematic review and cumulative meta-analysis of the primary outcomes of interest. This study was performed through a comprehensive search of multiple scientific databases, adhering strictly to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) and Assessing the Methodological Quality of Systematic Reviews (AMSTAR) guidelines. The literature search and screening process was conducted independently by two researchers. In the event of disagreements that could not be resolved through consensus, a third reviewer was enlisted to make the final determination. Four databases were searched: Embase, PubMed, the Cochrane Library, and Web of Science. The search strategy was designed to encompass three key concepts: (1) RCC, (2) AT, and (3) PN. These concepts were combined using the Boolean operator “AND.” Within each concept, relevant Medical Subject Headings (MeSH) and Emtree terms were utilized, along with a comprehensive list of free-text terms and synonyms (e.g., “RFA,” “cryoablation,” “nephron-sparing surgery”) connected by “OR” to maximize retrieval. The search was restricted to studies published in English. No other filters were applied. The search was restricted to studies published in English. No other filters were applied.
Eligibility Criteria
The following inclusion criteria will be applied for the selection of reports to be included in our systematic review: (1) the study subjects are diagnosed with renal cancer; (2) intervention (I): AT; (3) comparison (C): PN; (4) the study includes at least one of the following outcomes: operative time (OT), length of stay (LOS), estimated blood loss (EBL), and overall survival (OS), recurrence-free survival (RFS), CSS, estimated glomerular filtration rate (eGFR), complications, body mass index. The exclusion criteria are as follows: inability to extract relevant data; studies presented in the form of editorials, conference proceedings, or expert opinions; reports with overlapping study populations that present identical outcomes; studies involving non-human subjects; and studies that do not compare AT with PN. Systematic reviews and meta-analyses were excluded from quantitative data synthesis. These publications were consulted only for background information, contextual discussion, and identification of potential primary studies that might have been missed in the initial search, but data were never extracted from them for pooling.
Data Extraction
Data extraction was conducted by two independent reviewers who independently selected the articles to be included and extracted data in accordance with a pre-established data collection form. The data extracted comprised the following elements: author, year of publication, sample size, age, body mass index, tumor size, OT, length of stay (LOS), EBL, complications, eGFR, OS, RFS, CSS.
Study Quality Assessment
The Newcastle-Ottawa Scale (NOS) was employed to evaluate the quality of retrospective studies 9. The NOS scoring system is on a scale of 0–9, with a score exceeding 6 indicating high-quality studies.
Risk of Bias Assessment
The evaluation of the risk of bias within the included studies was independently undertaken by the same two authors.
Data Analysis
For the purpose of data analysis, we utilized the Stata version 16.0 software (StataCorp LLC, 4905 Lakeway Dr., College Station, TX, USA). In the context of our meta-analysis, the log relative risk along with its variance was employed as the summary outcome measure to synthesize information from all the trials under consideration. Specifically, for each individual trial, we calculated the hazard ratio and its corresponding 95% confidence interval (CI) for the survival rate. The choice between the fixed-effects model and the random-effects model was made based on the specific characteristics and data structure of each trial.
Statistical Analysis
The threshold for statistical significance was set at p < 0.05. WMD represented continuous variables, OR/relative risk represented dichotomous variables, and 95% CI were calculated. To assess the heterogeneity among the included studies, heterogeneity was assessed using the Q-test and the I2 statistic. If the I2 statistic exceeded 50% or the p value <0.10, it was interpreted as an indication of significant heterogeneity between the studies.
Results
Description of Study
The authors conducted a comprehensive search across four databases, yielding a total of 680 records. Utilizing Endnote software, they identified and removed 298 duplicate studies. A further 248 studies were excluded after reviewing their titles and abstracts. Additionally, 44 studies could not be retrieved, 32 were systematic reviews, 18 did not meet the inclusion criteria, and 8 were excluded due to incomplete data. Ultimately, 32 studies involving 6,030 patients were included in this meta-analysis. The sample sizes of these studies ranged from 27 to 2,276. All 32 studies were retrospective in nature. The screening process is illustrated in Figure 1, while the baseline characteristics of the included studies are detailed in Table 1. The 32 publications were released between the years 2007 and 2025. The PRISMA checklist is included in the online supplementary document (for all online suppl. material, see https://doi.org/10.1159/000550133).
Quality Assessment
The quality assessment of the cohort studies was performed utilizing the modified NOS, with the scores ranging from 6 to 8 points. A total of 32 studies were incorporated into this evaluation, and each of them achieved a score of at least six, as detailed in Table 2.
Operative Time
A total of 18 studies reported on the outcome of OT [12‒19, 26, 27, 30, 31, 35‒37, 39, 40]. Given the presence of substantial heterogeneity among these studies (I2 = 97.9%, p = 0.000), a random-effects model was employed for the meta-analysis. The pooled analysis revealed a significant difference between the groups with PN and AT (WMD = 76.70, 95% CI: [51.50–101.90], p < 0.05, I2 = 97.9%) (Fig. 2). PN is inherently more complex, leading to significantly longer OT compared to AT [43]. However, ablative technologies are continually advancing. In this study, we conducted subgroup analyses to compare different ablative methods (RFA, CA, MWA) and surgical approaches (LAT and PAT).
Subgroup analyses were conducted by the authors based on different ablative technologies. The pooled meta-analysis revealed a significant difference between PN and different AT (Fig. 3). The 5 studies [12‒16] show that the OT for PN is longer than that for RAF (WMD = 62.16, 95% CI: [12.72–111.61], p < 0.05, I2 = 96.4%).
The 5 results showed [15, 18, 19, 30, 31] that the OT of PN was longer than MWA (WMD = 78.30, 95% CI: [30.99, 125.60], p < 0.05). High heterogeneity between studies (I2 = 56.1%, p < 0.05, I2 = 98.8%) (Fig. 3). The 5 results [35‒37, 39, 40] showed that the OT of PN was longer than CA (WMD = 83.99, 95% CI: [8.2, 159.78], p < 0.05, I2 = 98.6%) (Fig. 3).
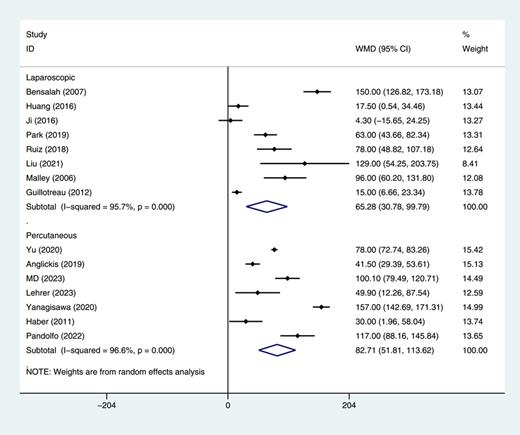
Subgroup analyses were conducted by the authors based on different surgical approaches (LAT and PAT). The pooled meta-analysis revealed a significant difference between PN and different surgical approaches of AT (Fig. 4). The 15 studies [12‒14, 16‒19, 26, 27, 35‒37, 39, 40] involving LAT and PAT have confirmed that the OT of AT is shorter compared to PN. LAT had a shorter OT compared to PN (WMD = 62.28, 95% CI: [30.78–99.79], p < 0.05, I2 = 95.7%). Additionally, another 7 studies also showed a significant difference in OT between PAT and PN (WMD = 82.71, 95% CI: [51.81–113.62], p < 0.05, 96.6%).
LOS (Days)
A total of 26 studies [12‒21, 23‒27, 30‒37, 39, 40] reported the length of stay (LOS). The pooled meta-analysis indicated significant difference in LOS between PN and AT (WMD = 2.01, 95% CI: [1.55–2.47], p < 0.05, I2 = 91.0%) as illustrated in Figure 5. Our findings suggest that AT is associated with shorter LOS compared to PN. This is likely due to the reduced trauma and quicker postoperative recovery associated with AT. To elucidate the differences in LOS across different ablative methods and surgical approaches, we conducted subgroup analyses.
The 10 results [12‒14, 16, 17, 20, 21, 23, 31, 32] showed that the LOS of PN was longer than RAF (WMD = 2.25, 95% CI: [1.68, 2.82], p < 0.05, I2 = 84.5%) (Fig. 6). The 4 results [15, 18, 19, 30] showed that the LOS of PN was longer than MWA (WMD = 3.66, 95% CI: [1.26, 6.07], p < 0.05, I2 = 88.6%) (Fig. 6).
The 7 results [33‒37, 39, 40] showed that the LOS of PN was longer than CA (WMD = 1.48, 95% CI: [0.81, 2.15], p < 0.05, I2 = 77.0%) (Fig. 6). The 25 studies [12‒14, 16‒21, 23‒27, 30‒37, 39, 40] involving LAT and PAT have confirmed that the LOS of AT is shorter compared to PN (Fig. 7). There was high heterogeneity among the eleven studies, which showed that LAT had a shorter LOS compared to PN (WMD = 1.53, 95% CI: [0.98–2.07], p < 0.05, I2 = 78.9%). Additionally, another 14 studies also showed a significant difference in LOS between PAT and PN (WMD = 2.25, 95% CI: [1.52–2.98], p < 0.05, I2 = 93.8%).
EBL (mL)
Estimated Glomerular Filtration Rate
Overall Complication Rate
Major Complication Rate
A total of 19 studies [13‒18, 22‒25, 27, 30, 31, 35‒37, 39, 41] reported the major complication rate (MCR) of PN vs. AT. Results showed that difference in MCR between PN and AT (OR = 1.73, 95% CI: [1.10–2.74], p > 0.05, I2 = 0.0%) (Fig. 11).
To explore the differences in MCR between different ablative methods and surgical approaches of AT versus PN, we conducted subgroup analyses. These seven findings show no significant difference in MCR between PN and RAF (OR = 1.78, 95% CI: [0.63, 4.99], p > 0.05, I2 = 32.1%) (Fig. 12). The 4 results show significant difference in MCR between PN and MWA (OR = 3.19, 95% CI: [1.19, 8.57], p < 0.05, I2 = 0.0%) (Fig. 12). The 5 results indicated that the MCR of PN was not significantly different from that of CA (OR = 1.04, 95% CI: [0.48, 2.23], p > 0.05, I2 = 0.0%) (Fig. 12).
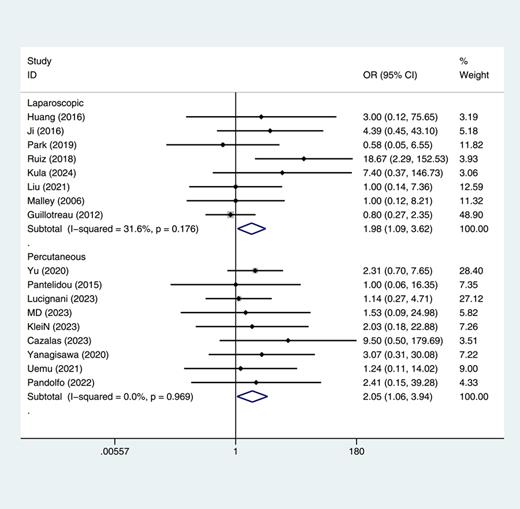
The 18 studies [13‒18, 22‒25, 27, 30, 31, 35‒37, 39, 41] involving LAT and PAT have confirmed that the MCR of AT is not significantly different from that of PN (Fig. 13). The 8 studies show significant difference in MCR between PN and LAT (OR = 1.98, 95% CI: [1.09–3.62], p > 0.05, I2 = 31.6%). Additionally, another 10 studies also showed significant difference in MCR between PAT and PN (OR = 2.05, 95% CI: [1.06–3.94], p > 0.05, I2 = 0.0%).
Overall Survival
The 10 studies [7, 14, 18, 19, 26, 28, 32, 39‒41] reported OS of AT vs. PN. Results showed that significant difference in OS between PN and AT (OR = 1.37, 95% CI: [1.13–1.66], p > 0.05, I2 = 34.7%) (Fig. 14).
This study performed subgroup analyses stratified by the different ablative methods, providing a clearer illustration of the OS differences between each ablation and PN. The 3 studies demonstrated that there was significant difference in OS between PN and RAF (OR = 1.69, 95% CI: [1.29, 2.22], p > 0.05, I2 = 0.0%) (Fig. 15).
The 3 studies demonstrated that there was no significant difference in OS between PN and MWA (OR = 1.19, 95% CI: [0.95, 1.51], p > 0.05, I2 = 43.8%) (Fig. 15). The 4 studies demonstrated that there was significant difference in OS between PN and CA (OR = 1.32, 95% CI: [0.68, 2.58], p > 0.05, I2 = 0.0%) (Fig. 15).
Our analysis indicates that the AT group does not have an advantage over the PN group in terms of OS. We anticipate that future well-designed clinical studies will provide more compelling evidence.
Recurrence-Free Survival
RFS is an essential time-to-event endpoint for assessing tumor treatment efficacy and prognosis, offering vital evidence for clinical decision-making and research design. Our pooled analysis of 15 included studies showed significant difference RFS between the PN and AT [7, 14, 15, 18, 22, 23, 26, 28, 29, 32, 38‒42] (OR = 1.32, 95% CI: [1.09–1.58], p > 0.05, I2 = 0.0%) (Fig. 16).
This study performed subgroup analyses stratified by the different ablative methods. The 5 studies demonstrated that there was no significant difference in RFS between RAF and PN (OR = 1.27, 95% CI: [0.69, 2.23], p > 0.05, I2 = 0.0%) (Fig. 17). The 4 studies demonstrated that there was no significant difference in RFS between PN and MWA (OR = 1.56, 95% CI: [0.75, 3.23], p > 0.05, I2 = 57.8%) (Fig. 17). The 4 studies demonstrated that there was significant difference in RFS between PN and CA (OR = 1.52, 95% CI: [1.03, 2.24], p > 0.05, I2 = 0.0%) (Fig. 17).
This study performed subgroup analyses stratified by the different surgical approaches. LAT was reported in 2 studies. The pooled meta-analysis demonstrated no significant difference in RFS between PN and LAT (OR = 0.92, 95% CI: [0.28–3.07], p > 0.05, I2 = 0.0%) (Fig. 18). PAT was reported in 9 studies. The pooled meta-analysis demonstrated significant difference in RFS between PN and PAT (OR = 1.45, 95% CI: [1.11–1.90], p < 0.05, I2 = 19.8%) (Fig. 18).
Cancer-Specific Survival
The 6 studies [7, 14, 18, 26, 39, 40] reported CSS of PN vs. AT. Results showed that no significant difference in CSS between PN and AT (OR = 1.37, 95% CI: [0.75–2.50], p > 0.05, I2 = 0.0%) (Fig. 19).
Our meta-analysis of 32 studies revealed that patients undergoing PN experienced longer OT, prolonged LOS, higher complication rate, and greater declines in postoperative eGFR. However, our analysis indicates that the AT group does not have an advantage over the PN group in terms of OS and RFS. These findings underscore the need for additional well-designed, high-quality studies to confirm these conclusions.
Discussion
The incidence of RCC continues to rise annually [1, 2]. Currently, nephron-sparing PN remains the standard treatment for small RCC [44]; nevertheless, AT represents a viable alternative for patients who are unable to tolerate PN [44‒46].
Our results demonstrate that AT offers significant advantages in several short-term outcomes. Specifically, AT was associated with shorter OT, reduced LOS, lower EBL, and better preservation of renal function, as reflected by higher postoperative eGFR. These benefits are consistent with the minimally invasive nature of AT, which avoids the complex dissection and reconstruction often required in PN [27, 45]. However, this advantage did not extend to long-term oncological outcomes.
A notable finding of this study is the considerable heterogeneity observed across multiple outcome measures, such as OT (I2 = 97.9%) and LOS (I2 = 91.0%). This variability likely arises from differences in surgical expertise, institutional protocols, patient selection criteria, and technological evolution over the study period [43]. Unlike previous meta-analyses [47, 48], this study conducted detailed subgroup analyses not only across ablation modalities (RFA, MWA, CA) but also between surgical approaches (laparoscopic vs. percutaneous). These analyses consistently demonstrated that AT – regardless of technique or access – was associated with shorter OT compared to PN. This difference likely stems from the inherently more complex nature of PN, which often requires renal vascular control and parenchymal reconstruction, whereas AT involves minimally invasive tissue destruction without the need for extensive dissection [27, 45].
The superior renal functional outcomes associated with AT are particularly noteworthy. The smaller decline in eGFR following AT is likely attributable to its tissue-preserving mechanism, which selectively destroys tumor tissue while minimizing damage to adjacent healthy parenchyma [49]. In contrast, PN inevitably removes a margin of normal kidney tissue, which may impair renal function to a greater extent [50]. Nevertheless, the clinical significance of this difference remains uncertain due to significant heterogeneity (I2 = 83.7%) and the limited number of studies reporting functional outcomes.
With regard to safety, our analysis revealed that PN did not offer advantages in terms of overall complications or major complications. This aligns with previous reports indicating that AT is a safe procedure with a morbidity profile comparable to that of surgery [8, 51]. Subgroup analyses further revealed no significant differences when AT modalities or approaches were considered separately, reinforcing the general safety of ablative techniques. However, variations in complication definitions and reporting standards across studies may have introduced additional heterogeneity, limiting the strength of this conclusion.
In the AT group, significant differences were observed in OS and RFS; nevertheless, these results must be interpreted with caution. The data suggest that AT is effective in controlling cancer-specific mortality, yet they may also reflect disparities in baseline patient characteristics – such as age and comorbidity burden – or technical factors including the completeness of ablation and the management of recurrences [52]. Notably, in contrast to earlier reports, a single-center study encompassing 1,798 patients demonstrated longer CSS with PCA than with PN, underscoring the potential influence of evolving surgical techniques [7]. Another recent retrospective study analysis also indicates that the risk of surgical-related complications for CA is extremely low [53]. These findings emphasize the imperative for meticulous patient selection and the establishment of standardized procedural protocols to optimize oncological outcomes.
Several limitations must be acknowledged. First, the predominance of retrospective studies introduces potential selection and reporting biases. Second, the high and largely unexplained heterogeneity across outcomes limits the robustness of our conclusions. Third, publication bias was not assessed, which may affect the validity of the results. Finally, rapid advancements in ablative technology mean that older studies may not reflect current practice.
Conclusion
AT demonstrates clear advantages over PN in perioperative and functional outcomes, supporting its role as a less invasive alternative for managing small renal masses. However, its association with inferior OS and RFS highlights the need for careful patient selection and long-term monitoring. Future prospective, well-designed studies are essential to better define the role of AT, standardize its application, and evaluate its long-term efficacy relative to surgical standards.
Acknowledgments
The original studies included in this meta-analysis were ethically reviewed. The author thanks Professor Zongping Zhang for his continued support and encouragement.
Statement of Ethics
A statement of ethics and consent to participate is not applicable because this study is based exclusively on published literature.
Conflict of Interest Statement
There are no conflicts of interest involved in this study.
Funding Sources
Zongping Zhang presided over the “Ten Thousand People Plan of Guozhou (ZX-W2201-4)” fund in Guizhou. Not only did he cover the expenses for article publication but he also participated in the design and revision.
Author Contributions
Ran Deng and Yunxiang Li participated in data analysis and validation. Zongping Zhang secured the funding for the research.
Data Availability Statement
The materials used in this study are openly available and can be accessed at PubMed, Embase, Cochrane, and Web of Science databases.