Abstract
Introduction: Mortality after radical cystectomy (RC) varies widely in the literature. In cohort studies, mortality rates can vary from as low as 0.5% in large-volume academic centers (2) to as high as 25% in developing countries series. This study aims to perform a systematic review of population-based studies reporting mortality after RC. Methods: A Systematic search was performed in Medline (PubMed®), Embase, and Cochrane for epidemiologic studies reporting mortality after RC. Institutional cohorts and those reporting mortality for specific groups within populations were excluded. Case series and non-epidemiologic series were also excluded. The aim of this review is to evaluate in-hospital mortality (IHM), 30-day mortality (30M), and 90-day mortality (90M). Results: Systematic search resulted in 42 papers comprising 449,661 patients who underwent RC from 1984 to 2017. Mean age was 66.1. Overall IHM, 30M, and 90M were 2.6%, 2.7%, and 4.9%, respectively, with 90M being 2.6 times higher than IHM on average. Lowest IHM was found in Canada and Australia (0.2% and 0.6%, respectively), while the highest IHM was 7.8% (Brazil). Canada and Spain showed the highest 90M (6.5%). 159,584 urinary diversions were analyzed, being mostly ileal conduits (76.8%). Conclusions: The majority of the studies available are from major developed economies with paucity of data in the developing world. 90M after RC tends to be at least twice as high as IHM. The knowledge of such epidemiologic data is vital to guide public policies, such as centralization, in order to reduce mortality.
Introduction
Radical cystectomy (RC) is currently considered as the standard of care for patients with muscle-invasive bladder cancer (MIBC) without systemic involvement and, less commonly, for some nonmuscle-invasive bladder cancer, mainly after failure of intravesical treatments. It is advocated as the mainstream treatment for most patients with MIBC [1]. RC is, however, associated with considerable postoperative complications and mortality. Mortality after RC varies widely in the literature. In cohort studies, mortality rates can vary from as low as 0.5% in large-volume academic centers [2] to as high as 25% in developing countries series [3, 4]. Cohort studies, however, have several biases, including sampling bias (e.g., small cohorts have results associated with a particular institution/surgeon experience), selection bias (e.g., referral centers operate mostly on patients with excellent surgical prognosis), and publication bias (e.g., only favorable results are published). There is not also a clear definition in the literature about the best way to report mortality after RC. While some studies report in-hospital mortality (IHM), others report 30-day mortality (30M) or 90-day mortality (90M) rates. Despite the wide variation of the mortality rates in the literature, previous studies have highlighted a pattern among populational studies with IHM being lower than 30M, which in turn is two- to three-fold lower than 90M [5]. Large analytic nonexperimental population-based epidemiological series can mitigate some of these aforementioned biases posed by cohort studies. Therefore, the present study aims to perform a systematic review of all available epidemiologic studies reporting mortality after RC.
Patients and Methods
Evidence Acquisition and Search Strategy
The search was performed using Medline (Pubmed®), Embase, Cochrane following the standards set by Preferred Reporting Items for Systematic Review and Meta-Analyses statement (PRISMA) [6]. These databases were searched systematically for all population-based studies accessing mortality after RC using the terms cystectomy AND population-based OR factual database* OR “Surveillance, Epidemiology, and End Results (SEER) program” published up to January 1, 2020. The search was limited to the English, Spanish, Portuguese, French, and German literature. References were manually reviewed by two investigators (F.K. and W.B.) to identify supplementary studies of interest.
Definitions
IHM comprises deaths that have occurred before hospital discharge, and 30M comprise deaths occurring within 30 days. 90M represents all-cause mortality within the first 90 days after RC.
Eligibility Criteria
All studies presenting mortality rate after RC were included. The population selected was patients diagnosed with bladder cancer (BC) and submitted to RC, regardless of type of urinary diversion. The resulting articles were searched for data IHM, 30M, and 90M. Only noninstitutional cross-sectional studies were considered to avoid reporting bias.
Any institutional study, even when it was a cross-sectional study, was excluded. Articles that evaluated specific groups within populations (e.g., extremely elderly) were excluded. Other exclusion criteria were non-humans’ studies, case series, non-epidemiologic series, epidemiologic series not reporting surgical mortality. On full-text review, studies that appeared to be duplicate analyses of the same cohorts of data were also excluded.
Study Selection and Data Collection Process
Two paired investigators (F.K. and W.B.) independently screened all articles focusing the research on population-based studies with epidemiologic information on MIBC, mainly mortality related to RC. Discrepancies about eligibility were solved by a discussion between the two investigators, and, if necessary, a third researcher (F.T.) was consulted until consensus was reached.
After full-text selection, demographic data (age, sex, country), type of urinary diversion (ileal conduit [IC], neobladder, cutaneous ureterostomy [CU]), staging, IHM, 30M, and 90M were collected and analyzed. Studies were aggregated by country to create larger data samples for analysis.
Data Quality Assessment
Two investigators (F.K. and F.T.) independently assessed the risk of bias using the Cochrane tool for “risk of bias” assessment and followed as described in the Cochrane Handbook for Systematic Reviews of Interventions [7]. All authors approved a priori protocol (International Prospective Register of Systematic Reviews, ID number 181554).
Results
Evidence Synthesis
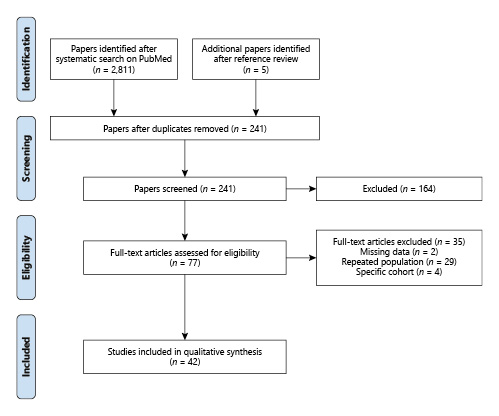
The study selection is presented in Figure 1. [3, 8‒46] A total of 42 studies comprising 449,661 patients who underwent RC from 1984 to 2017 were evaluated. About 75% of the patients were men (n = 337,802) and 25% (n = 111,859) were women. The mean age of patients was of 66.1 years (60–75 years).
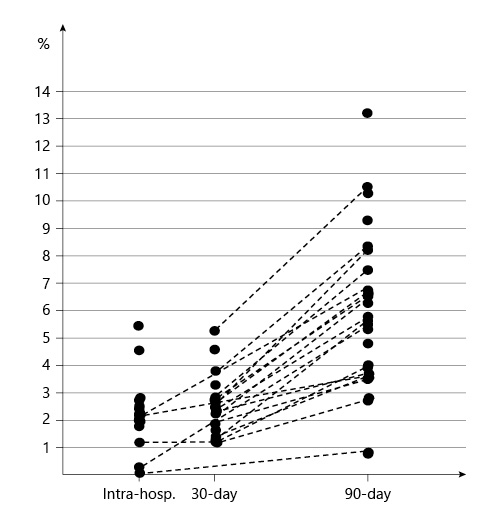
Overall IHM, 30M, and 90M mortality rates were 2.6%, 2.7%, and 4.9%, respectively. Mean mortality rates were chosen to be reported as the vast majority of the papers selected did not include data about median mortality rates. On average, 90M rate was 2.6 times higher than IHM, ranging from 1.6 to 27.5 times higher than IHM (Fig. 2). Figure 2 demonstrates the ratio between mortality at distinctive moments when reported by the same study.
In-Hospital Mortality Rate
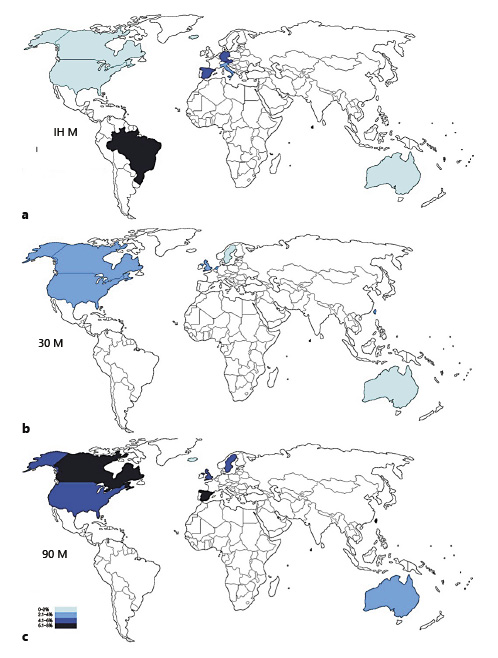
IHM was reported in 19 studies comprising 347,953 patients and ranged from 0 to 7.8% (mean 2.6%). Björnsson et al. [10] reported the lowest IHM (zero), among patients who have undergone RC in Iceland from 2003 to 2012 (n = 108), mostly males (81%), and with a median age of 68 years. About 40.7% of the patients bear a disease with pathological stage > pT2 and 16.6% N+. The preferred urinary diversion was IC 86%, followed by orthotopic neobladder 14%. Timoteo et al. [3] reported the highest IHM (7.8%) among 5,091 patients in Brazil. Figure 3 summarizes mean IHM in each country where population-based studies were available (online suppl. Table 1; see www.karger.com/doi/10.1159/000524578 for all online suppl. material).
30-Day Mortality Rate
30M rate was reported in 18 studies (85,106 patients) and ranged from 1.1% to 9.3% (mean 2.7%). Isbarn et al. [25] reported the lowest 30M (1.1%) among 5,510 patients with a median age of 67.1 years. Patients were mostly male (74.7%); pathological staging and urinary diversion techniques were not mentioned.
The highest 30M (9.3%) was found in a study that used the United States Renal Disease System Database to identify patients treated with RC between 1984 and 2013 that were on hemodialysis by the time of surgery (n = 985) [28]. About 68.5% of the patients did not receive any urinary diversion due to end-stage renal disease. Figure 3 illustrates mean 30M in each country.
90-day Mortality Rate
90M rate was reported in 25 studies (167,762 patients) and ranged from 0.9% to 13.1% (mean 4.9%). The lowest 90M was demonstrated by Björnsson et al. [10] among 108 patients in Iceland; this study’s population characteristics were shown above. Dell’Oglio et al. [15] reported the highest 90M rate (13.1%) among 1,000 patients in the USA, treated with RC between 2000 and 2009. About 70% were male, and the median age was 75.2 years [15]. Figure 3 illustrates mean 90M in each country where epidemiologic studies were available.
Mean National Mortality Rates
When aggregated by country, Canada and Australia stand out with the lowest IHM rates, 0.2%, and 0.6%, respectively. The highest IHM is 7.8% in Brazil. Minimum and maximum 30M rates are observed in Sweden (1.2%) and the Netherlands (3.2%). The highest 90M rates are found in both Canada and Spain (6.5%), whereas in Iceland, this rate drops to 0.9% (online suppl. Table 1).
Urinary Diversion
In some of the series, the urinary diversion procedure was reported. IC was the most common procedure reported (63.0%–88.8%). Continent diversions (CD) were reported in up to 37% in Sweden. CU was also reported in some of the series, representing up to 11.9% of Italy’s procedures. When a pooled analysis was performed, 159,584 of the procedures could be evaluated. Of the total, IC was performed in 76.8% (n = 119,805), CD in 10.4% (n = 16,252), CU in 0.02% (n = 24), and in 12.8% (n = 20,012), the urinary diversion was not mentioned (online suppl. Table 2).
In most studies, mortality rates were not reported for all postoperative periods. Only two studies reported IHM, 30M, and 90M [14, 35].
Discussion
There is a lack of epidemiologic studies evaluating mortality and outcomes after RC. In the USA and Canada, several databases allow the evaluation of these epidemiologic studies. We have obtained many of the studies from databases such as the SEER, National Cancer Database, US Renal Disease System Database, American College of Surgeons’ National Surgical Quality Improvement Program, National Inpatient Sample, Nationwide Readmissions Database, Texas Hospital Discharge Public Use Database, State of Washington Comprehensive Hospital Abstract Reporting System, Premier Healthcare Database, Régie de l’Assurance Maladie du Québec, ISQ, MED-ÉCHO, Canadian BC Network Database, Ontario Cancer Registry. In other countries such as England, Sweden, Australia, Iceland, Germany, Italy, the Netherlands, Taiwan, and Brazil, only a limited number of databases allowed evaluation of mortality. The gathering of data regarding almost half a million patients who underwent RC obtained from these epidemiologic series brought some critical insights.
Finding an “Unbiased” Mortality Rate
First, we could estimate the mortality rates for a vast number of patients who underwent RC. Mean IHM and 90M were 2.6% and 4.9%. These numbers are much higher than those reported in a controlled scenario. In an extensive series of more than 5,000 selected patients from high-volume academic institutions in the USA who underwent RC, a meager mortality rate of 0.5% was observed [2]. It is essential to know these numbers to establish treatment strategies, to inform patients better, and to improve outcomes in centers worldwide.
Second, there was an extensive variation in mortality rates in epidemiologic series. In such complex procedures, there are several known factors associated with each particular health system that can either improve or be detrimental to the results. These include access to the health system, stage at diagnosis, nutrition status, age, and comorbidities. In this setting, it is essential to highlight the impact of centralization of care [47]. Waingankar et al. [48] have demonstrated that patients who undergo RC in a low-volume facility (<5 RC/year) are exposed to a 60% increase in 30M and 40% increase in 30M when compared to those treated in a high-volume institution (>30 RC/year).
Establishing an “Early-Late Mortality Ratio”
Alongside bleeding and infections, there are many additional factors, including thromboembolic events, cardiovascular events, bowel complications, and wound dehiscence that might impact mortality after RC. For this reason, early IHM rates cannot be evaluated solely. The IHM definition has been criticized for being dependent on the length of stay and discharge practices. The expansion of fast-track programs for cancer surgery affirms the relevance of collecting postdischarge data to provide valid estimates of 30M. The 30M definition is therefore recommended as an international standard because it includes the vast majority of surgery-related deaths and is not subject to discharge procedures. Long-term postoperative mortality might also be assessed, and for such a purpose, 90M seems to be a reasonable indicator. The 90M definition captures mortality from multiple causes, including clinical conditions.
In the present study, mean IHM was 2.6%. After 90 days, the mean mortality rate increased to 4.9%. Our findings suggest that 90M was, on average, twice as common as IHM (Fig. 2). Our group has previously demonstrated that this 90M/IHM ratio seems to be around 2–3 in most cohort studies [5]. This ratio can be used as an additional tool to help predict expected mortality outcomes.
Lack of Data from Developing Countries
The only data available regarding countries with developing economies are from Brazil and Taiwan and they accounts for 5.4% of the individuals included in the analysis. The majority of data available (94.6%; 425,306 patients) come from countries classified by the United Nations as major developed economies [49]. Even though we did not perform statistical analysis to compare the groups, IHM was higher in Brazil than in other countries. In the few large cohorts of RC from developing countries, it is not uncommon to observe mortality rates as high as 25% [4]. Unfortunately, there were no data available regarding low-income countries, as classified according to the UN level of development classification.
It is known that mortality rates are expected to be higher in lower income countries. Disparities in mortality can be seen even when developed countries are compared to more developed countries. A study in Europe evaluating postoperative mortality for all surgical procedures demonstrated a wide variation, with an overall IHM rate varying from 0.44 in Finland to 6.92 in Poland [50].
When health issues are evaluated in low-income countries, however, these disparities become much more critical. For appendectomy, for example, the mortality rate in the USA is approximately 3 per 1,000 surgeries [51]. For the same procedure, in lower-middle-income countries, like Nigeria, the appendectomy mortality rate is 6.3, in South Africa 12.5, and in Congo 17.9. In low-income countries, this scenario becomes catastrophic, with mortality rates after appendectomy being 40 per 1,000 procedures in Ethiopia and 88.6 per 1,000 in the Central African Republic [52]. If we can observe this wide mortality variation after an appendectomy, we can expect much higher mortality for more complicated procedures. In an epidemiologic study in Brazil, our group has observed IHM rates around 4.3% after RC in the Southern and more developed regions of the country. In contrast, in one of the northern and least developed states of the country, the IHM rate rises to 38% [3].
Even though there are no epidemiologic data available from RC in developing countries, there are some cohorts of RC published, and some of them express these concerns. In developing countries, reported IHM rates are 9.6% in Mexico [53], 6.9% in India [54], 4.5% in Pakistan [55] and 30M are 8.5% in Brazil [56], 5% in Saudi Arabia [57], and 23% in South Africa [4]. When analyzing 90M in developing countries, reported rates are 17.3% in Chile [58] and 23% in Egypt [59]. Alarmingly, when applying the 2–3× factor observed in the present study, estimated 90M rates for these cohorts would be somewhat between 9.0% and 46%.
It is well known that countries in northern Africa have elevated incidence of squamous carcinoma of the bladder, which is a histological variant of BC that commonly affects younger patients. Unfortunately, our systematic research did not result in any eligible epidemiologic studies from these countries, and therefore, mortality could not be assessed in this specific group of patients.
Real-World RC Population
Current guidelines on BC recommend that patients with pT2-pT4 M0 disease undergo RC [1]. However, most of the patients presenting with these characteristics are not treated according to these guidelines. As exposed in a study based on the SEER-Medicare database, only 18.9% of the patients with MIBC were managed with RC [60]. This finding suggests that in large cohorts, there is a selection of the patients who undergo RC and those who are treated with chemotherapy, radiotherapy, join clinical trials, or are offered best supportive care. In the present review, we found that the mean age for operated patients was 66.1 years. The mean age in the distinctive series ranged from 60.4 to 75.0 years. These patients seem to be relatively young. BC is a disease that increases in frequency with age. According to our findings, however, it appears that the patients with BC treated with RC are mainly the younger and healthier. Even though there are some series of elderly patients treated with RC published [12], it seems to be the exception according to these large epidemiologic cohorts. Most elderly patients with MIBC might be treated worldwide through alternative procedures [60]. This finding follows the increment of radiotherapy being applied solely but mainly as a multimodality treatment for these patients.
Urinary diversions performed vary widely according to patients’ health, preference, surgeons’ experience, and many other factors. Although it was not the primary purpose of the present study, we found that the IC remained the most common diversion in this epidemiologic series. CD was performed in 10.4% of the patients. In patients from Swedish Bladder BaSe, the Hospital Discharge Records of Northern Italy, and the SEER in the USA, a CD was performed in up to one-third of the patients. On the other hand, in different cohorts such as in the North-American National Inpatient Sample database, IC was performed in the vast majority of patients (online suppl. Table 2). It is noteworthy that even in the studies in which the diversion was mentioned, in a significant number of cases (12.2%, n = 19,052), the type of diversion was unknown.
Horizons to Improve Outcomes
It has been previously demonstrated that a sophisticated system of postoperative care is essential in managing complications and preventing them from resulting in death. Although surgical complication rates were similar across the USA, mortality rates varied dramatically after major procedures in distinctive hospitals, suggesting that the main difference relies on the failure to rescue these patients when complications occur [61].
Complications after RC are common and expected [62, 63]. An experienced surgeon in a high-volume and high-quality hospital will have the ability to recognize promptly, intervene, and ultimately prevent these complications that result in death [42, 64, 65]. Several effective strategies have been identified for improving outcomes following RC. Centralization programs have been adopted in most of the countries that evaluated their epidemiologic series of RC, and these centralization programs have proved to be effective in reducing mortality [9, 66]. In England, centralization programs for RC are associated with a 40% decline in 30M [66].
The present study has some limitations and biases that cannot be accounted for, and therefore, our results should be interpreted with caution. The data in the distinctive databases are heterogeneous. It is also impossible to assure that some patients are not duplicated in different databases.
Furthermore, the retrospective nature of the studies evaluated in our study limits the precision of the information. Missing data of specific series may also present a limitation. Due to missing data, we cannot be certain about disease staging and preoperative care in all series, which could be specially interesting when comparing mortality outcomes between developing and major developed countries. However, the massive number of patients comprised in the 42 studies included increases the precision and allows generalizability of predictions.
Conclusion
Mortality rates after RC vary widely across hospitals and regions of the world. RC 90M tends to be at least twice as high as IHM. Further understanding of factors contributing to these wide discrepancies in mortality rates is essential to improve the assistance of these patients. The knowledge of such epidemiologic data is vital to set reference parameters and to allow public policies such as centralization programs to improve outcomes for such complex procedures.
Statement of Ethics
Ethical approval was not required for this study in accordance with local/national guidelines.
Conflict of Interest Statement
The authors have no conflicts of interest to declare.
Funding Sources
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author Contributions
Conception and design: Dr. Fernando Korkes. Acquisition of data: Dr. Willy Baccaglini and Dr Felipe Glina. Statistical analysis and interpretation of data: Dr. Fernando Korkes, Dr. Frederico Timóteo, and Dr. Oscar Rodriguez Faba. Drafting of the manuscript: Dr. Frederico Timoteo and Dr. Fernando Korkes. Critical revision of the manuscript for important intellectual content: Dr. Fernando Korkes, Dr. Juan Palou, and Dr. Sidney Glina.
Data Availability Statement
All data generated or analyzed during this study are included in this article and its supplementary material files. Further inquiries can be directed to the corresponding author.